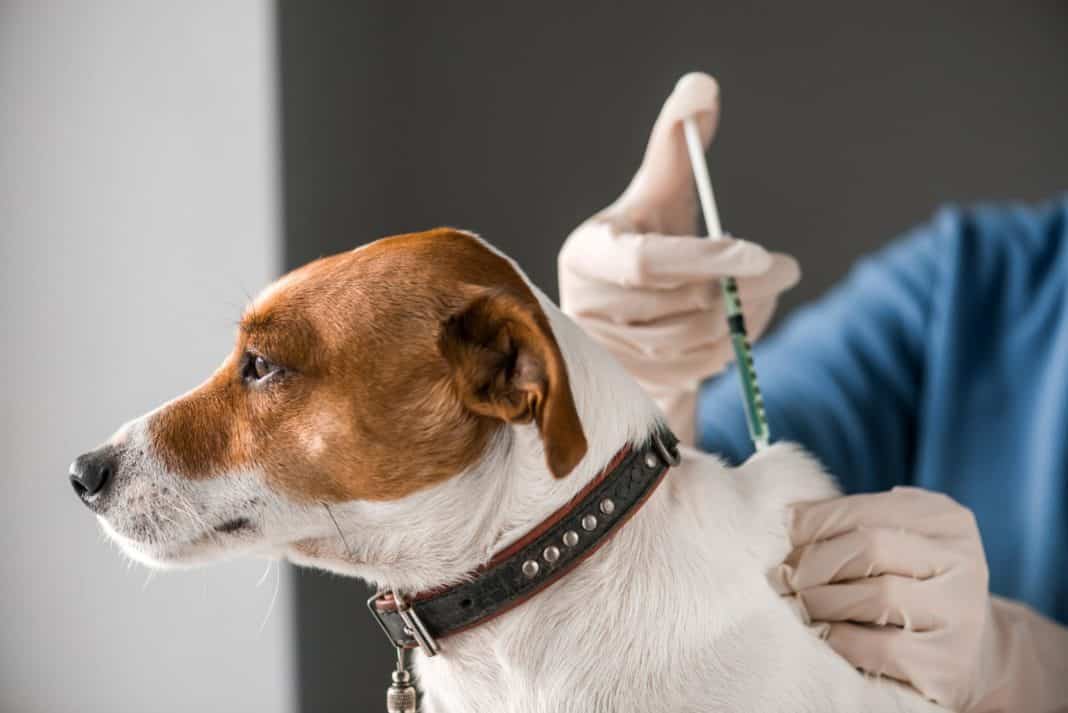
Clinical trials of the Carnivac-Cov vaccine has involved dogs, cats, foxes, mink and other animals, Russia’s agriculture safety watchdog has said
Russia has said that it has registered the world’s first COVID-19 vaccine for animals.
Clinical trials of Carnivac-Cov started in October and involved dogs, cats, foxes, mink and other animals, the country’s agriculture safety watchdog Rosselkhoznadzor said on Wednesday.
The vaccine was developed by a unit of the agency, which said immunity lasts for six months after vaccination but this is continuing to be analysed by the jab’s developers.
“The results of the trials allow us to conclude that the vaccine is harmless and highly immunogenic as all the vaccinated animals developed antibodies to the coronavirus in 100% of cases,” said the regulator’s deputy head Konstantin Savenkov.
this comes after the Pfizer-BioNTech COVID-19 vaccine is safe and 100% effective in children as young as 12, its manufacturers have said. The two firms have carried out vaccine trials in the US on 12 to 15-year-olds, which they say were successful.
“It is the world’s first and only product for preventing COVID-19 in animals,” he added.
Mass production of the vaccine could start as early as April, the watchdog said. It also claimed the jab would help prevent mutations in animals.
There have been reports of three pets diagnosed with COVID-19 in Mexico, one in Italy, and two cat infections in Russia
There is no scientific proof that the novel coronavirus might be transmitted from animals to humans, Mr Savenkov said.

In November, millions of mink were culled after a COVID mutation was found at more than 200 Danish mink farms.
The drastic action was taken after a “unique variant” of SARS-CoV-2 – the virus that causes COVID-19 – was discovered in 12 people infected by minks.
Russia already has three coronavirus vaccines for humans, the most well-known of which is Sputnik V.
This comes after Boris Johnson has joined over 20 other world leaders in calling for a new global settlement to help the world to prepare for future pandemics. In a newspaper article the leaders, including the German chancellor and French president, said COVID posed the biggest challenge since World War Two.
Leading medical journal The Lancet said last month Sputnik V was safe and 91.6% effective against coronavirus when two doses are given, following interim results of its phase three trial.
The preliminary findings were based on analysis of data from nearly 20,000 participants – three-quarters of whom received the vaccine with the rest given a placebo.
Moscow has also given emergency approval to two other vaccines, EpiVacCorona and CoviVac.